 Determine the electron group arrangement around the central atom that minimizes repulsions.Draw the Lewis electron structure of the molecule or polyatomic ion.This VESPR procedure is summarized as follows: VESPR Produce to predict Molecular geometry The Valence Shell Electron Pair Repulsion model was developed to translate these 2-dimensional drawings into 3-dimensional shapes which illustrate the bond. #Vsepr shapes how to#Using this information, we can describe the molecular geometry, the arrangement of the bonded atoms in a molecule or polyatomic ion. This lesson plan includes the objectives, prerequisites, and exclusions of the lesson teaching students how to identify the shape of molecules using VSEPR. valence-shell electron-pair repulsion (abbreviated VSEPR and. From the BP and LP interactions we can predict both the relative positions of the atoms and the angles between the bonds, called the bond angles. geometry, or three-dimensional shape of a molecule or polyatomic ion, can be determined using. Each group around the central atom is designated as a bonding pair (BP) or lone (nonbonding) pair (LP). 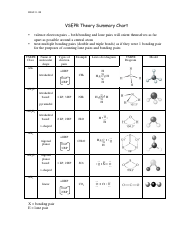
The acronym 'VSEPR' is pronounced 'vesper' for ease of pronunciation. 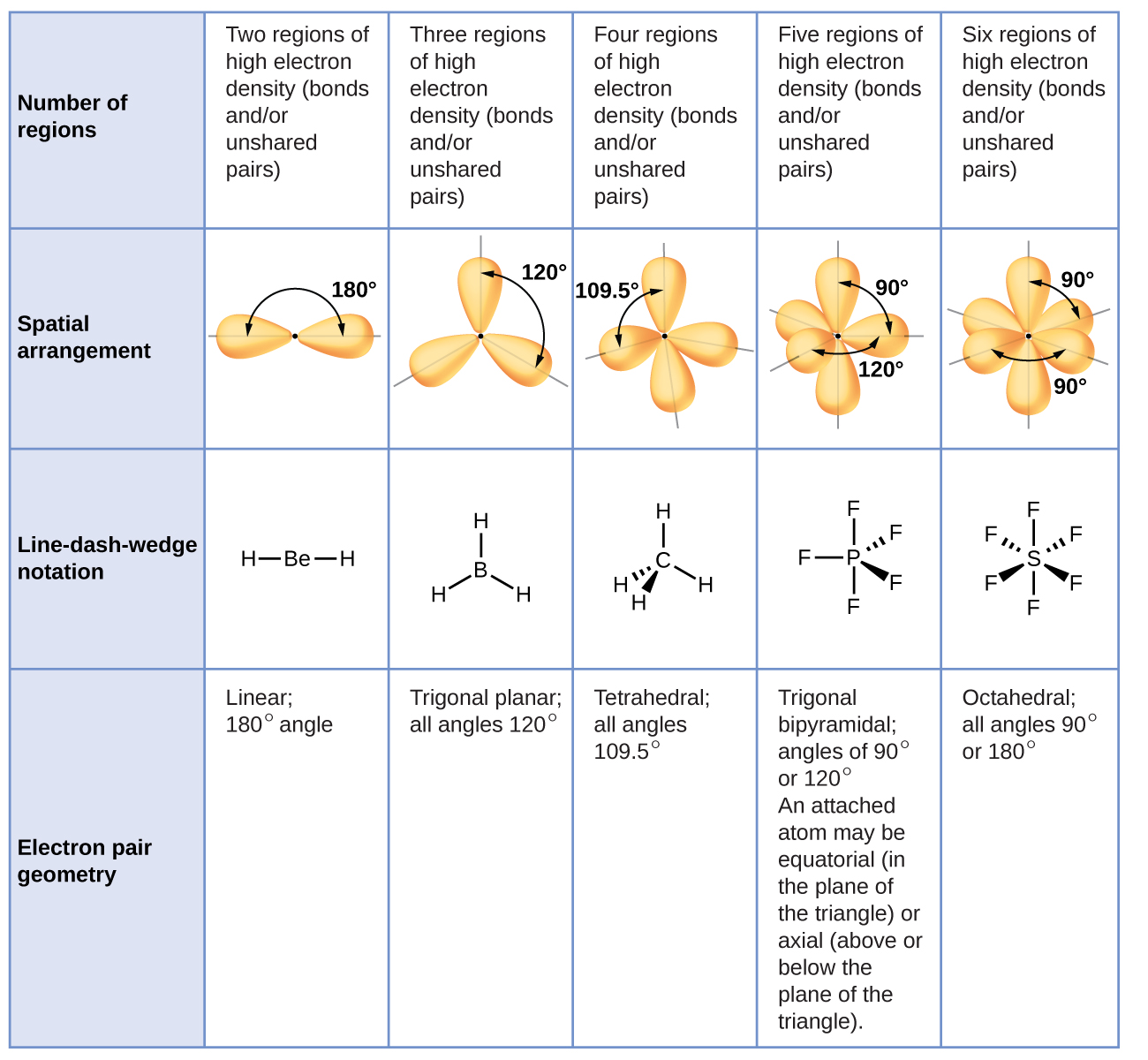
It is also named Gillespie-Nyholm theory after its two main developers. In the VSEPR model, the molecule or polyatomic ion is given an AX mE n designation, where A is the central atom, X is a bonded atom, E is a nonbonding valence electron group (usually a lone pair of electrons), and m and n are integers. Valence shell electron pair repulsion (VSEPR) theory is a model in chemistry used to predict the shape of individual molecules based upon the extent of electron-pair electrostatic repulsion. 3D VSEPR app helps you to visualize the shapes of the VSEPR models in 3D such that you can. 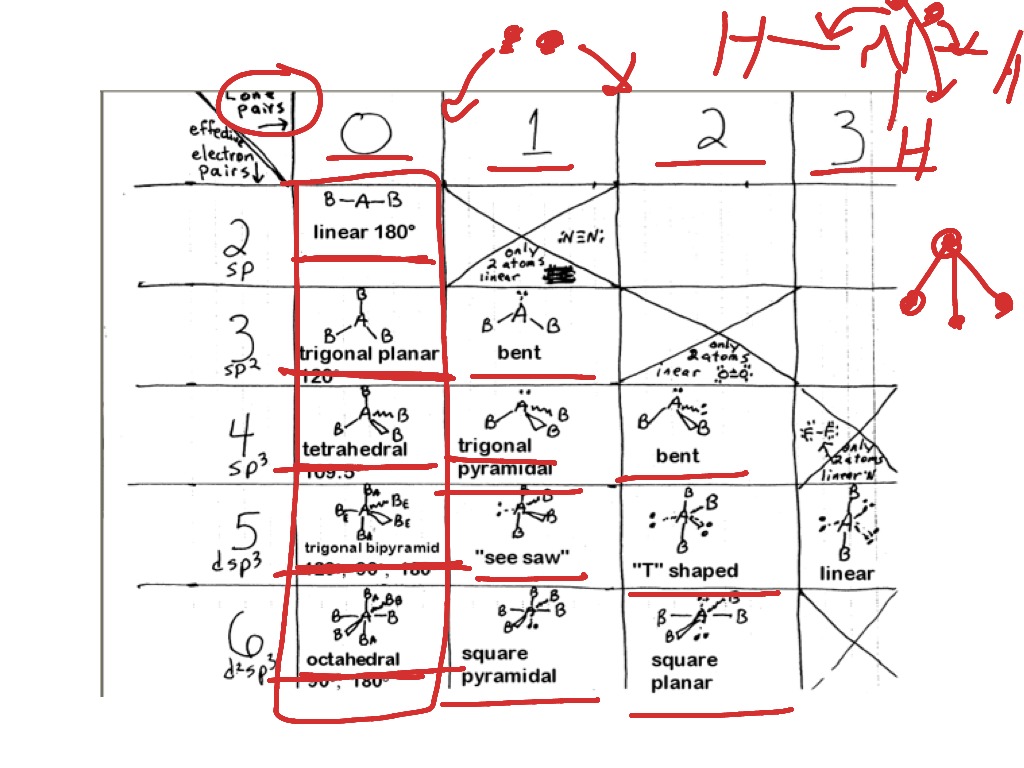
Groups are placed around the central atom in a way that produces a molecular structure with the lowest energy, that is, the one that minimizes repulsions. It is impossible to learn about the shapes of VSEPR models in a 2D page because these arent in 2D. \): Electron Geometries for Species with Two to Six Electron Groups. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
